Among the most devastating consequences of spinal cord injury is the sudden silence between the brain and the body. A person may retain thought, intention, and the will to move—yet the neural pathway that carries those signals is damaged. For decades, such injuries were considered permanent barriers to movement. Today, spinal cord stimulation technology challenges that assumption. Spinal cord stimulators represent one of the most promising intersections of neuroscience, bioengineering, and rehabilitation medicine.
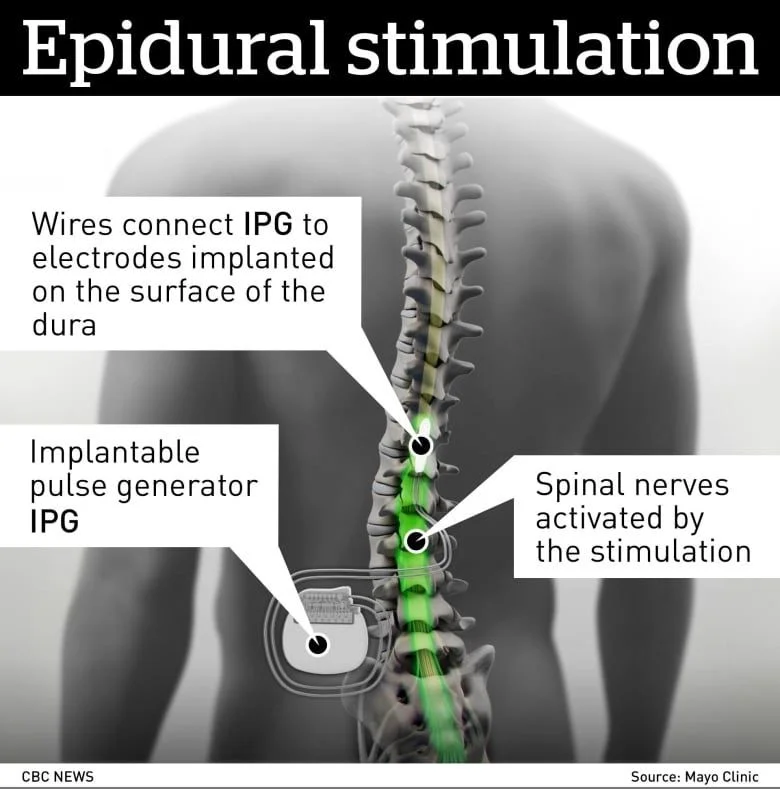
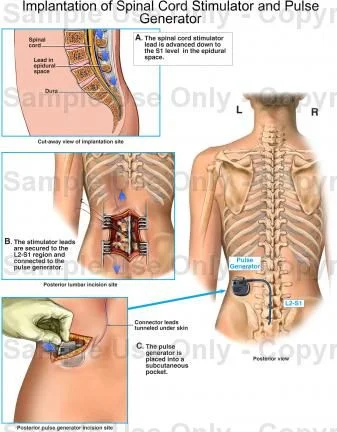
A spinal cord stimulator (SCS) is an implanted medical device that delivers controlled electrical pulses to specific regions of the spinal cord. These pulses are generated by a small programmable unit placed under the skin, typically in the abdomen or lower back. Thin insulated wires, known as leads, carry electrical signals to electrodes positioned near the spinal cord. By stimulating targeted nerve circuits, the device can alter pain signals or, in emerging applications, reactivate dormant motor pathways.
Originally developed in the 1960s and 1970s to treat chronic pain, spinal cord stimulation was based on the “gate control theory” of pain. According to this theory, electrical stimulation can interrupt pain signals before they reach the brain. Over time, SCS became a widely used treatment for conditions such as failed back surgery syndrome and neuropathic pain. However, recent research has expanded its purpose beyond pain management into the realm of motor recovery.
In patients with spinal cord injuries, communication between the brain and muscles is disrupted, but the spinal cord below the injury often retains some functional neural circuits. Electrical stimulation can “awaken” these circuits. By applying patterned pulses that mimic natural neural activity, spinal cord stimulators enhance the excitability of motor neurons, enabling voluntary movement in some cases. Clinical studies in recent years have demonstrated that individuals with partial paralysis can regain the ability to stand, take assisted steps, or improve muscle control when stimulation is combined with intensive rehabilitation.
The mechanism behind this recovery is both electrical and biological. Stimulation does not rebuild the spinal cord physically; instead, it amplifies residual neural signals and encourages neuroplasticity—the brain and spinal cord’s ability to reorganize and form new connections. In some groundbreaking experiments, researchers have paired spinal cord stimulators with brain–computer interfaces to create a “digital bridge” between the brain and spinal circuits. These systems interpret movement intentions from brain signals and translate them into spinal stimulation patterns, enabling more coordinated stepping motions.
Despite these advances, spinal cord stimulation is not a cure. Its effectiveness depends on the type and severity of injury, and many patients require ongoing therapy and device adjustments. Surgical risks, hardware complications, and long-term maintenance remain considerations. Ethical questions also arise regarding access, cost, and equitable distribution of advanced neurotechnology.
Nevertheless, spinal cord stimulators symbolize a profound shift in medical philosophy. Rather than accepting paralysis as an irreversible condition, modern medicine increasingly views it as a communication problem that may be modulated through electrical intervention. The spinal cord is no longer seen solely as a passive transmission cable but as an active neural network capable of adaptation and reactivation.
From a broader perspective, spinal cord stimulation reflects the rise of “electroceuticals”—medical treatments that use targeted electrical impulses instead of drugs. As understanding of neural circuits deepens, electrical therapies may become more personalized and precise. Future developments may include closed-loop systems that automatically adjust stimulation in real time, guided by sensors that monitor muscle activity or neural feedback.